Example of a primary drying design space graph showing sublimation
4.6 (246) In stock
4.6 (246) In stock

PDF) Recommended Best Practices for Lyophilization Validation—2021

Recommended Best Practices for Lyophilization Validation-2021 Part I: Process Design and Modeling. - Abstract - Europe PMC
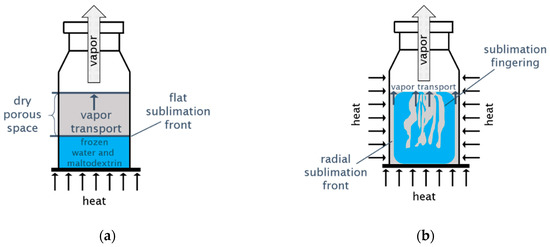
Processes, Free Full-Text

PDF) Recommended Best Practices for Lyophilization Validation—2021

Optimization of Primary Drying by Construction of a Graphical Design Space Video

Recommended Best Practices for Lyophilization Validation-2021 Part

Figure A1. Product temperature for 5.4 mL and 10.6 mL fill volumes

Choked flow and importance of Mach I in freeze-drying process

Best Practices and Guidelines (2022) for Scale-up and Technology Transfer in Freeze Drying Based on Case Studies. Part 2: Past Practices, Current Best Practices, and Recommendations

Ehab MOUSSA, Senior Scientist, PhD, AbbVie, Illinois

Tong ZHU, Senior Scientist I, Ph.D. in Aero Eng.

The graphical design space for the primary drying phase of freeze Drying: Factors affecting the dried product layer resistance - ScienceDirect
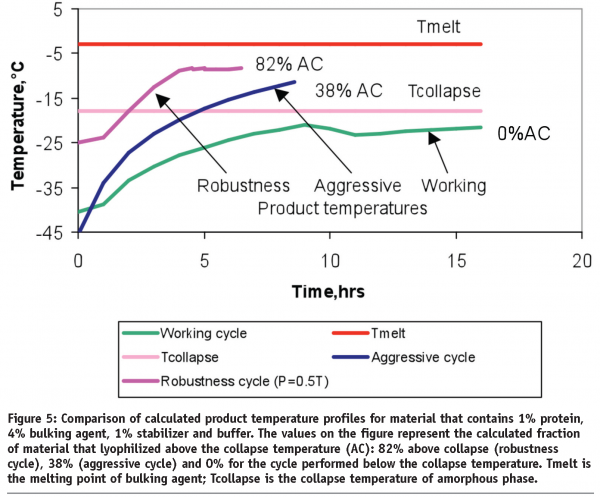
Lyophilization: cycle robustness and process tolerances, transfer and scale up - European Pharmaceutical Review

Ehab MOUSSA, Senior Scientist, PhD, AbbVie, Illinois

Feroz Jameel's research works Gilead Sciences, Foster City and