
the compression factor one mole of a vander waals gas 0 C and 100
4.7 (621) In stock

4.7 (621) In stock
Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor for one mole of a vander waals gas at 0 c and
Click here👆to get an answer to your question ✍️ The compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0-5

The compression factor (compressibility factor) for one mole of a Van der..

29. The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate the volume of a 4 mole sample of Co, same temperature and pressure (use R = 0.08 L. atm/K.mol (
How I find the a and b constant in the Van der Waals equation? - Quora
How I find the a and b constant in the Van der Waals equation? - Quora

For one mole of a Vander Waals gas when b = 0 and T = 300 K, the PV vs 1/V plot is shown below. The value of the van der Waals

The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is

123. The compra compressibility factor one mole of a der Waals gas 0º C and 100 atm pressure found to be 0.5 Assume that the volume of es molecule is negligible. The

What is the compressibility factor (Z) for 0.02 mole of a van der Waals's gas at pressure of 0
The value of compression factor at the critical state of a vander waals gas is
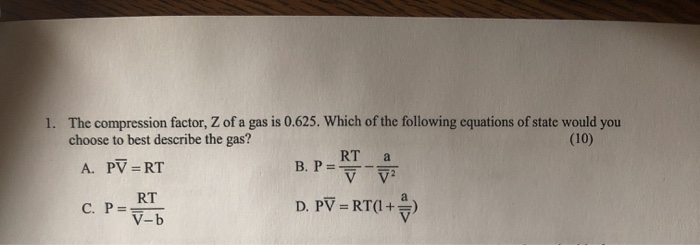
Solved 1. The compression factor, Z of a gas is 0.625. Which

The compression factor (compressibility factor) for 1 mol of a van der
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect
How can we calculate critical temperature, volume and pressure in terms of a and b? - Quora