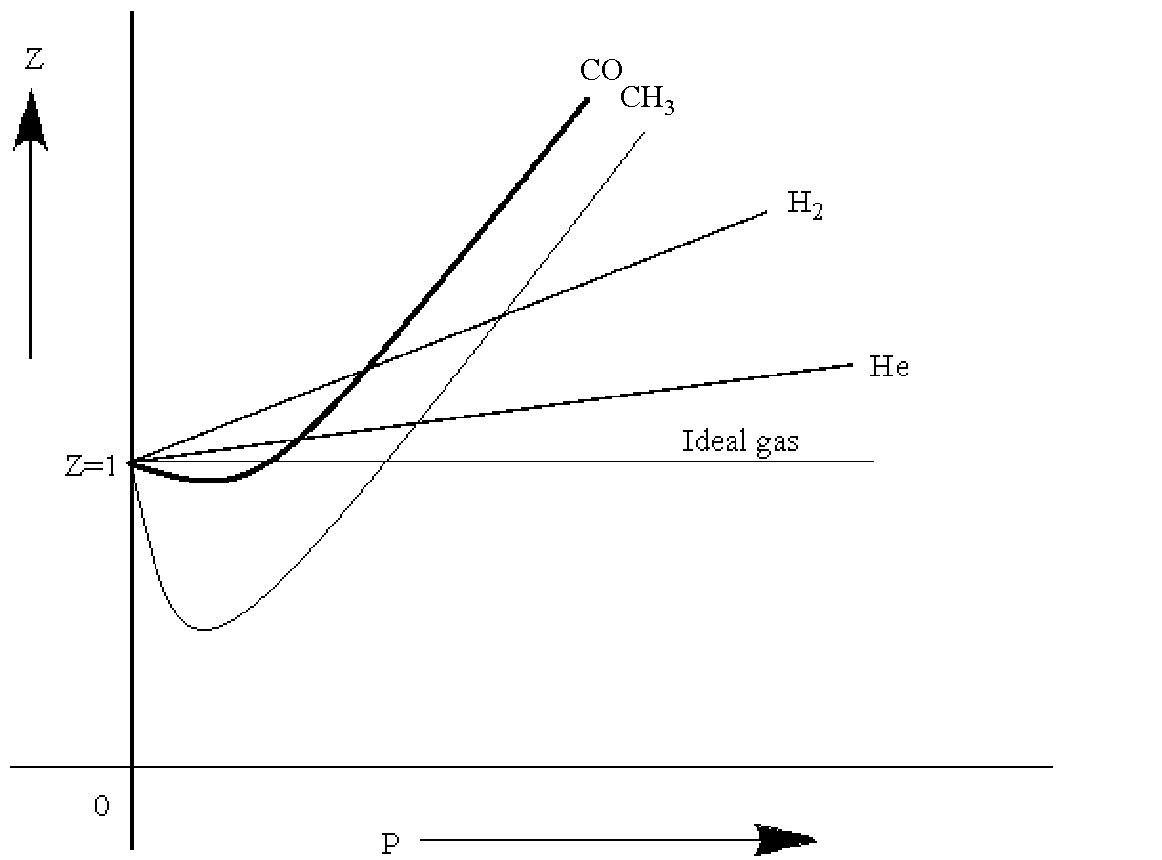
The graph of compressibility factor (Z) vs. P for one mole of a real gas ..
4.5 (580) In stock
4.5 (580) In stock
Solution For The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following diagram. The graph is plotted at constant temperature 273K. If the slope of graph at very hi
The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following diagram. The graph is plotted at constant temperature 273K. If the slope of graph at very high pressure (dPdZ) is (2.81)atm−1 , then calculate volume of one mole of real gas molecules (in L/mol) Given : NA=6×1023 and R=27322.4LatmK−1mol−1

REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR

Compressibility Factor of Gas Overview, Equation & Chart

Deviation From Ideal Gas Behavior - Study Material for IIT JEE

VMC - SOSE JEE MAINS - 4 (Question Paper), PDF, Gases

Compressibility factor (gases) - Citizendium
Boyle's temperature or Boyle point is the temperature at which a

VMC - SOSE JEE MAINS - 4 (Question Paper), PDF, Gases
Is z (compressibility factor) vs P (pressure) graph drawn by
What is compressibility factor (Z)? - Sarthaks eConnect

Compressibility factor z versus 100/V, for several values of