
Preparation of Standard Solution of Sodium Carbonate - Chemistry
4.6 (206) In stock

4.6 (206) In stock
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU'S to understand more about it.
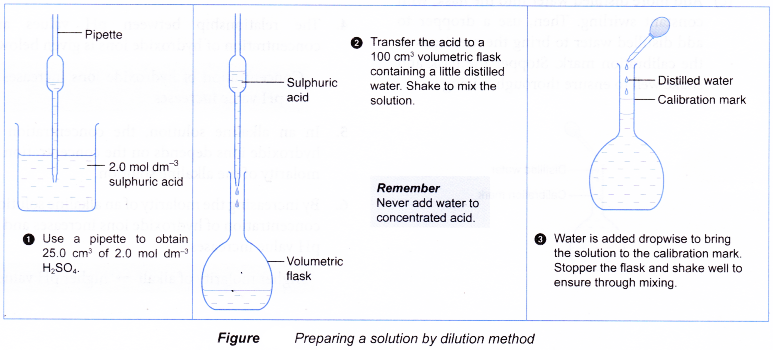
How do you prepare a standard solution? - A Plus Topper

Preparation of Standard Solution of Sodium Carbonate - Chemistry

Preparation and standardization of various molar and normal

Preparation and standardization of 1N HCl solution

PREPARATION OF 0.1 N SODIUM CARBONATE SOLUTION.

Discussion For Titration and Preparation of Standard Solution

Sodium Carbonate - an overview

Standard Solution-Sodium Carbonate

Calculating molarity units molar concentration of solutions

PREPARATION OF SODIUM CARBONATE BY SOLVAY PROCESS