
Calculate the number of protons, electrons, neutrons in Bromine with atomic number 35 and mass number 80.
4.5 (133) In stock

4.5 (133) In stock
Click here:point_up_2:to get an answer to your question :writing_hand:calculate the number of protons electrons neutrons in bromine with atomic number 35 and mass
Click here👆to get an answer to your question ✍️ Calculate the number of protons- electrons- neutrons in Bromine with atomic number 35 and mass number 80

Calculate the work, in J, if the volume of a system contract
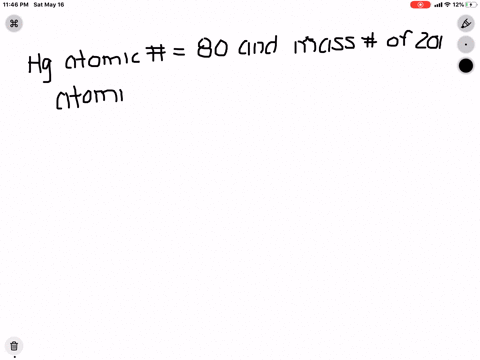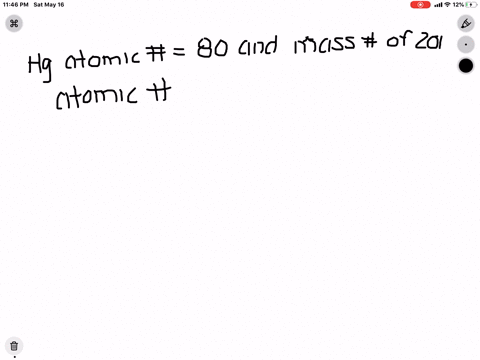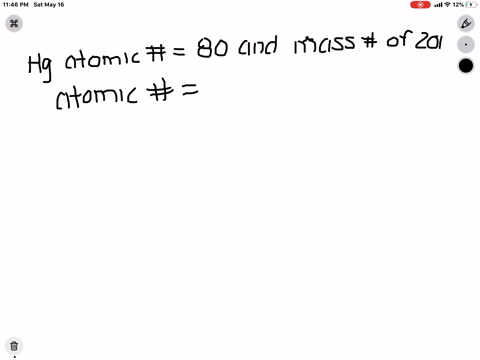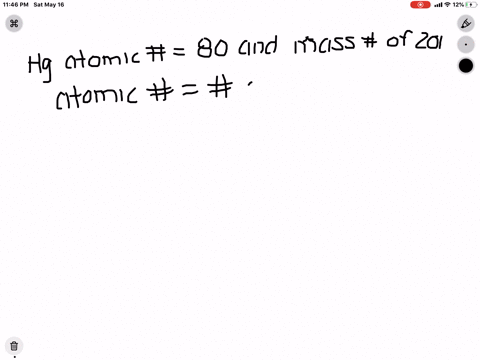
SOLVED: What is the atomic number, mass number, number of electrons, number of protons, and number of neutrons of Bromine-80

Calculate the number of protons, neutrons and electrons in 8035 Br.

4 Go The atomic masses of two isotopes of O are 15.9936 and 17.0036, calculate in each atom q) number of neutrons, o number of protons, number of electrons, d) mass number.
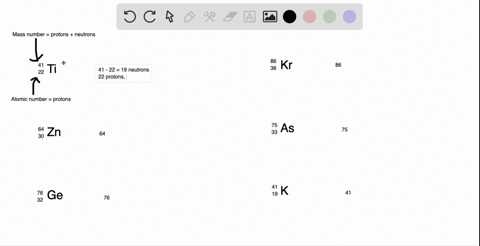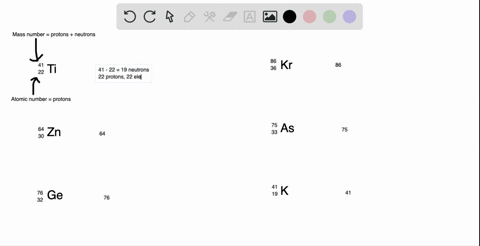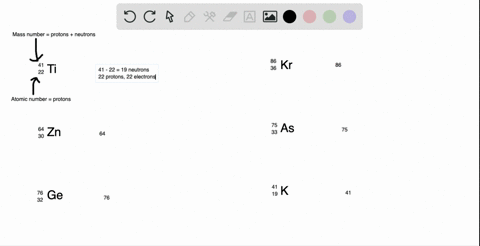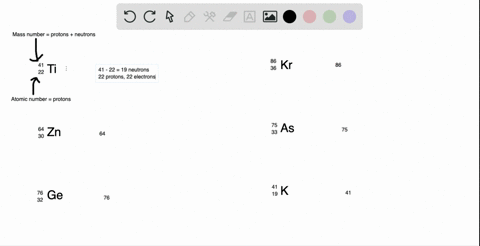
SOLVED: What is the atomic number, mass number, number of electrons, number of protons, and number of neutrons of Bromine-80

Calculate the number of protons, electrons, neutrons in Brom
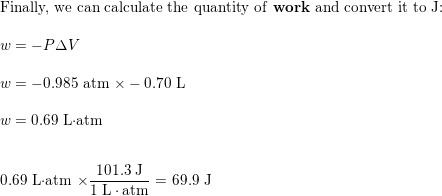
Calculate the work, in J, if the volume of a system contract

Calculate the number of protons, electrons, neutrons in Brom
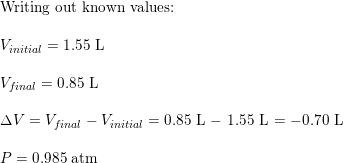
Calculate the work, in J, if the volume of a system contracts from 1.55 to 0.85 L at a constant pressure of 0.985 atm.

Calculate the work, in J, if the volume of a system contract