
At a high pressure, the compressibility factor (Z) of a real gas is us
4.8 (616) In stock

4.8 (616) In stock
At high P. P gt gt (n^(2)a)/(V^(2)) So ‘a’ can be neglected.

Determine Compressibility of Gases

JEE: Van der Waals Equation, Chemistry By Unacademy

At a high pressure, the compressibility factor (Z) of a real gas is usual..

3.2 g of oxygen gas is placed in a vessel of 10 litre at 1000 K so tha

Compressibility factor - Wikipedia

Compressibility factor (z): real gases deviate from ideal behav-Turito
At Critical Temperature,pressure and volume . The compressibility Factor (Z) Is

PDF] Natural Gas Compressibility Factor Measurement and Evaluation for High Pressure High Temperature Gas Reservoirs
The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT - Sarthaks eConnect

Express the average kinetic energy per mole of a monoatomic gas of mol
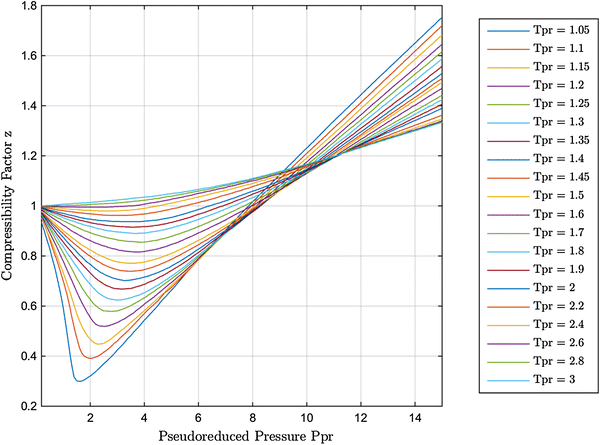
New explicit correlation for the compressibility factor of natural gas: linearized z-factor isotherms