Ranking The Bulkiness Of Substituents On Cyclohexanes: A-Values
4.6 (650) In stock

4.6 (650) In stock
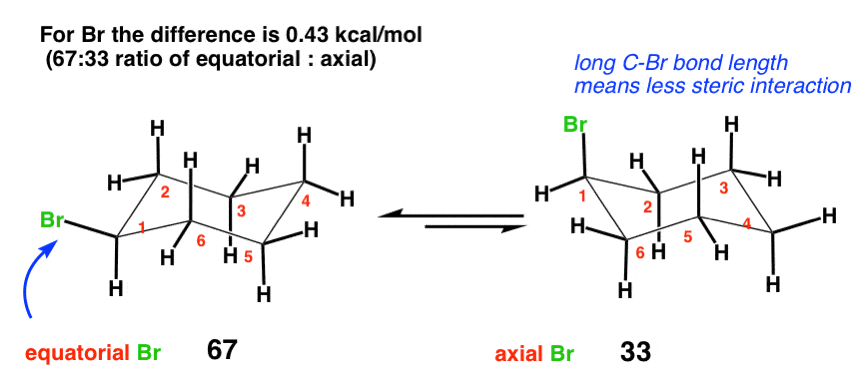
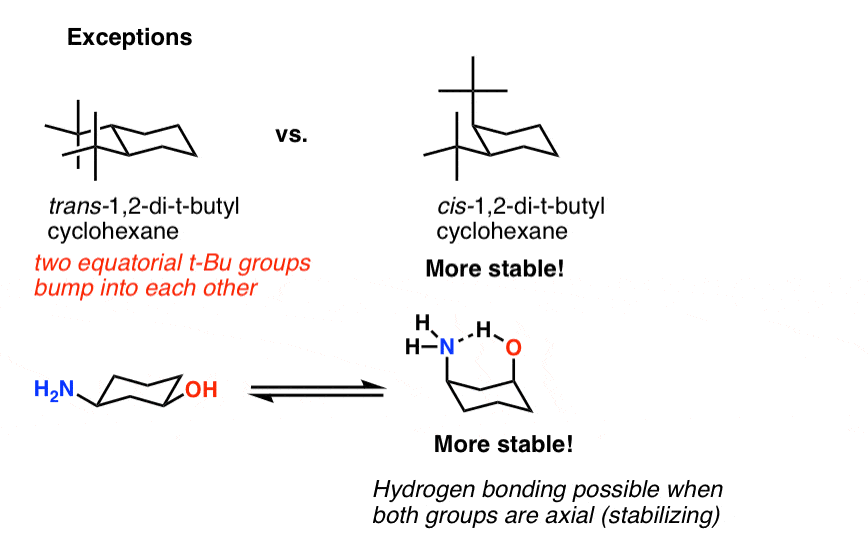
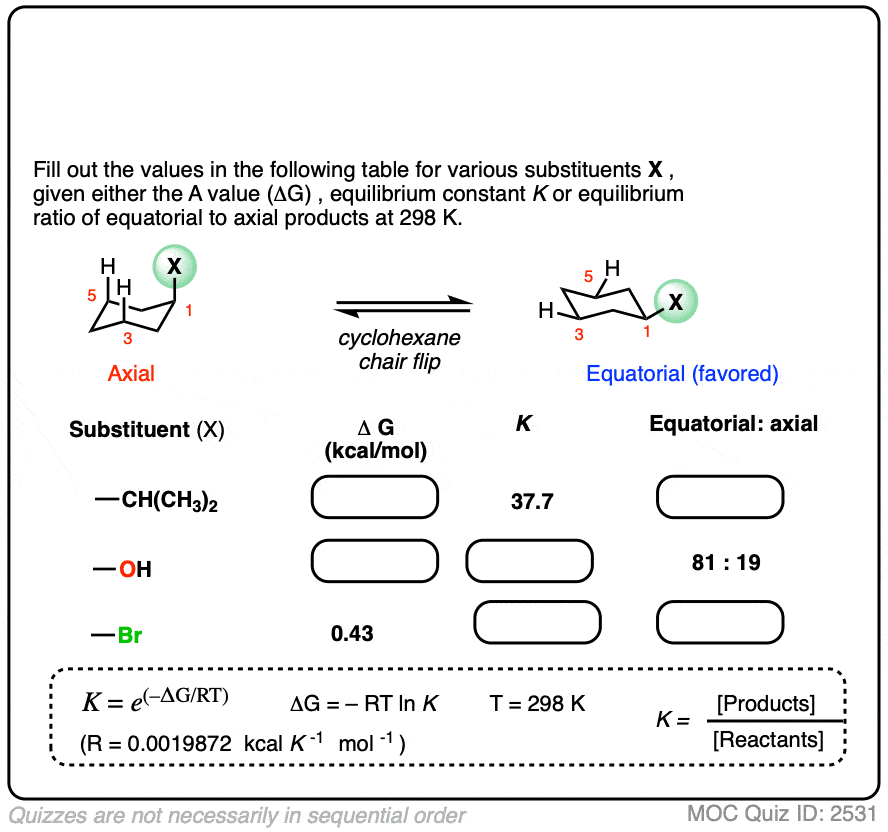
The "steric influence" (or "bulk") of groups on a cyclohexane is quantified by measuring the equilibrium btw axial and equatorial. This is the "A-value"
Cyclohexane Chair Conformation Stability: Which One Is Lower Energy?

Monosubstituted Cylcohexanes
4.1: Cyclohexane Ring Conformations - Chemistry LibreTexts

3.12: Substituted Cycloalkanes - Chemistry LibreTexts
Equilibrium and Energy Relationships – Master Organic Chemistry
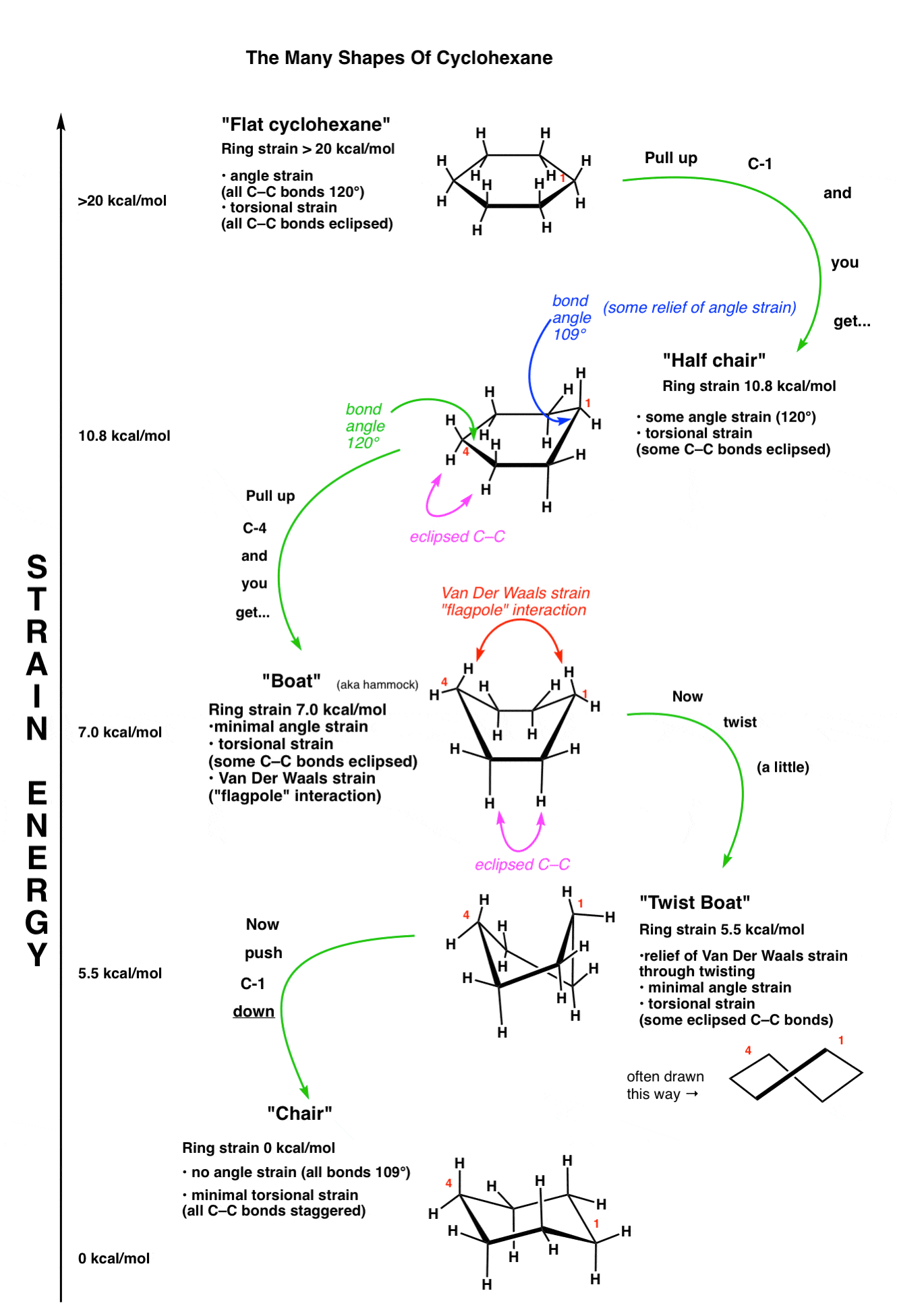
Cyclohexane Conformations – Master Organic Chemistry
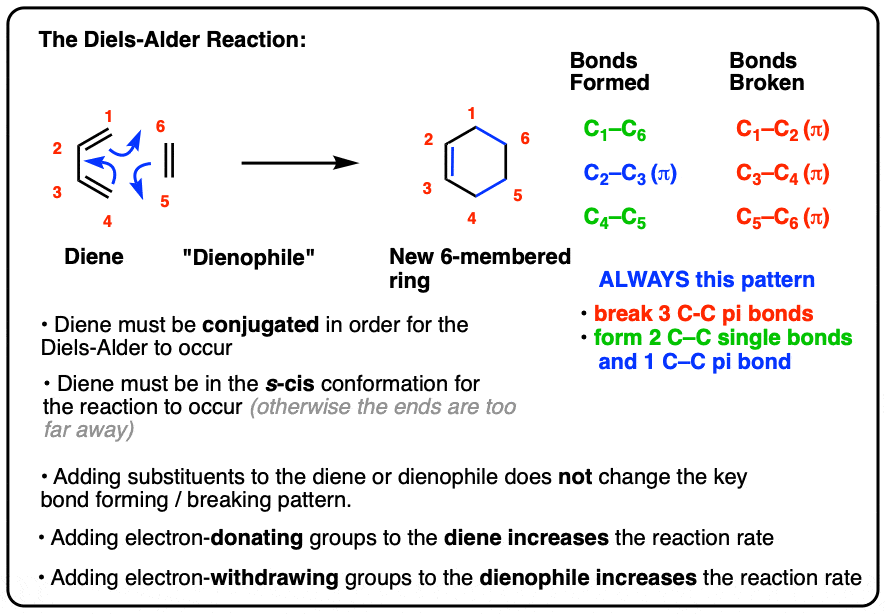
The Diels-Alder Reaction – Master Organic Chemistry
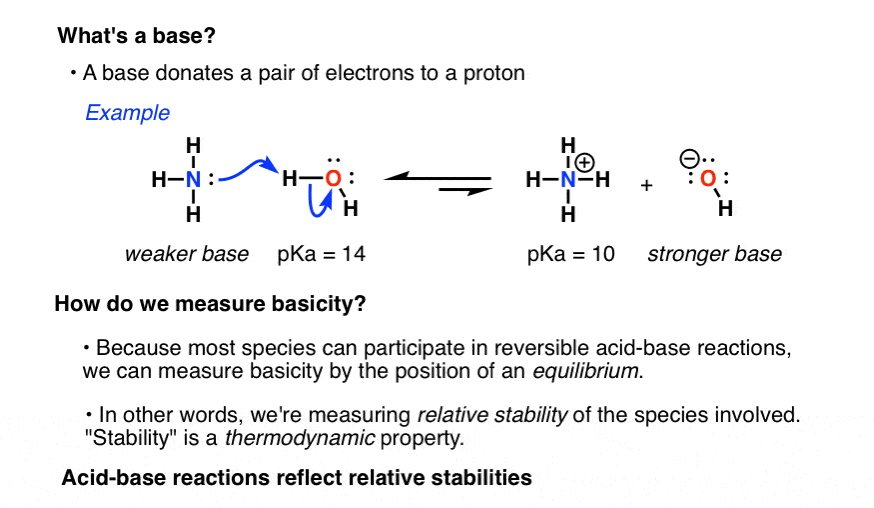
Nucleophilicity vs. Basicity – Master Organic Chemistry
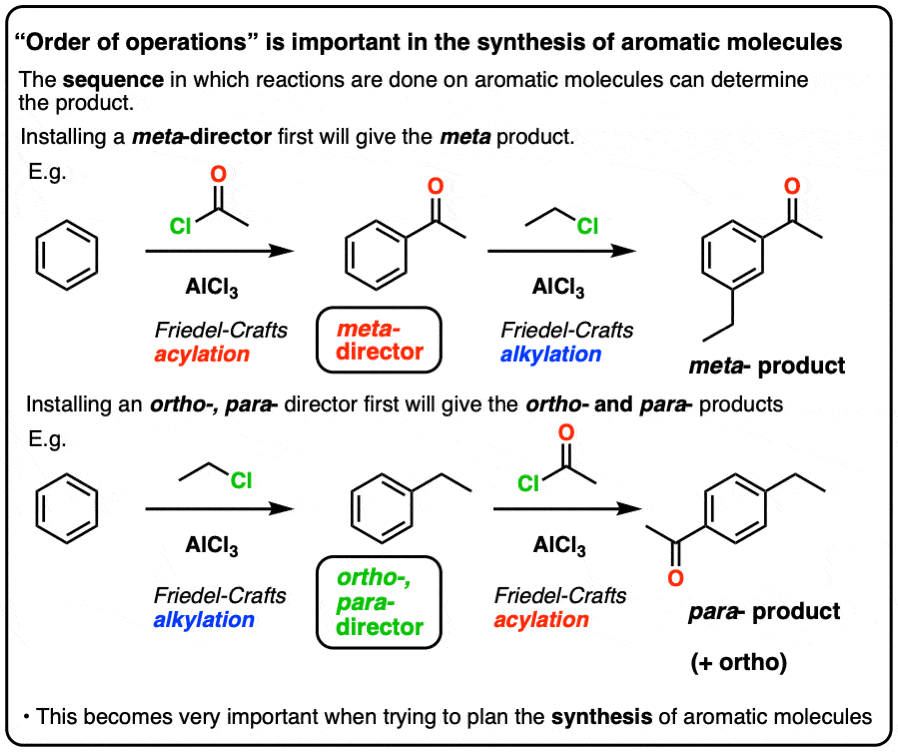
Aromatic Synthesis: Order of Reactions - Master Organic Chemistry

Polar substituent effect of the ester group on conformational equilibria of O-mono-substituted cyclohexanes—the para-substituent effect in cyclohexyl benzoates - ScienceDirect
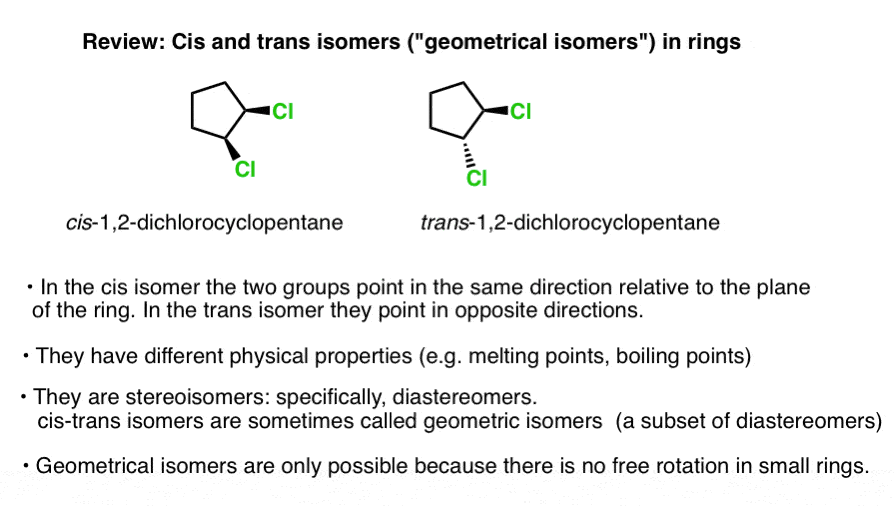
E and Z Notation For Alkenes (+ Cis/Trans) - Master Organic Chemistry

Calculating Energy Difference Between Chair Conformations - Video Tutorials & Practice Problems
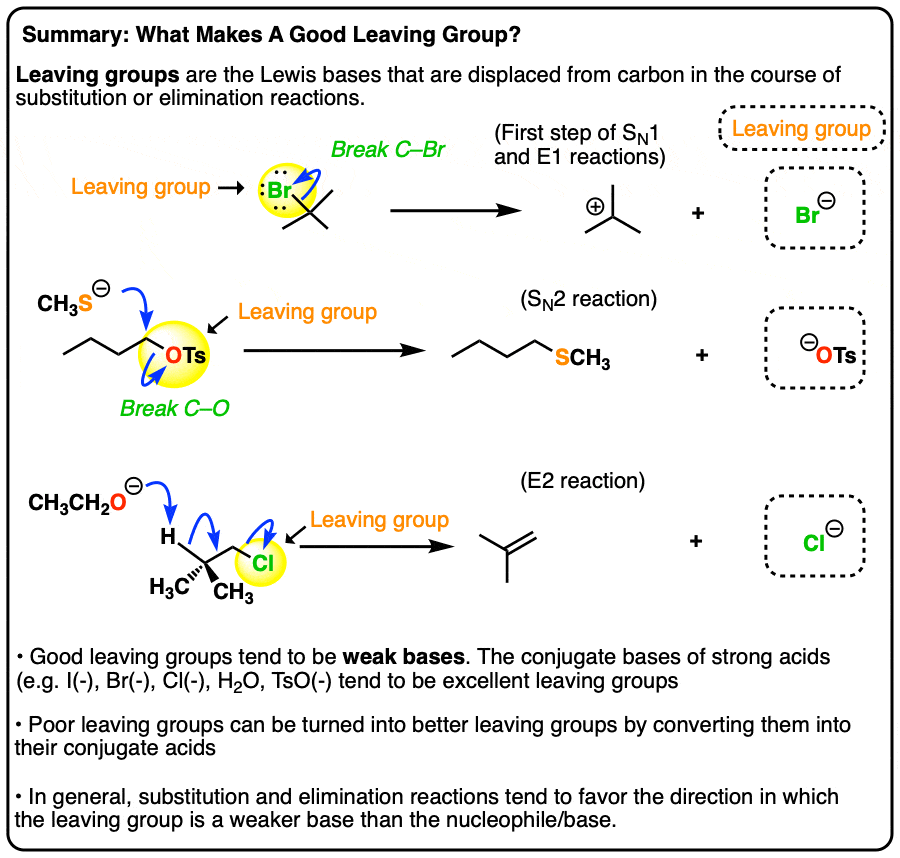
What makes a good leaving group? Master Organic Chemistry
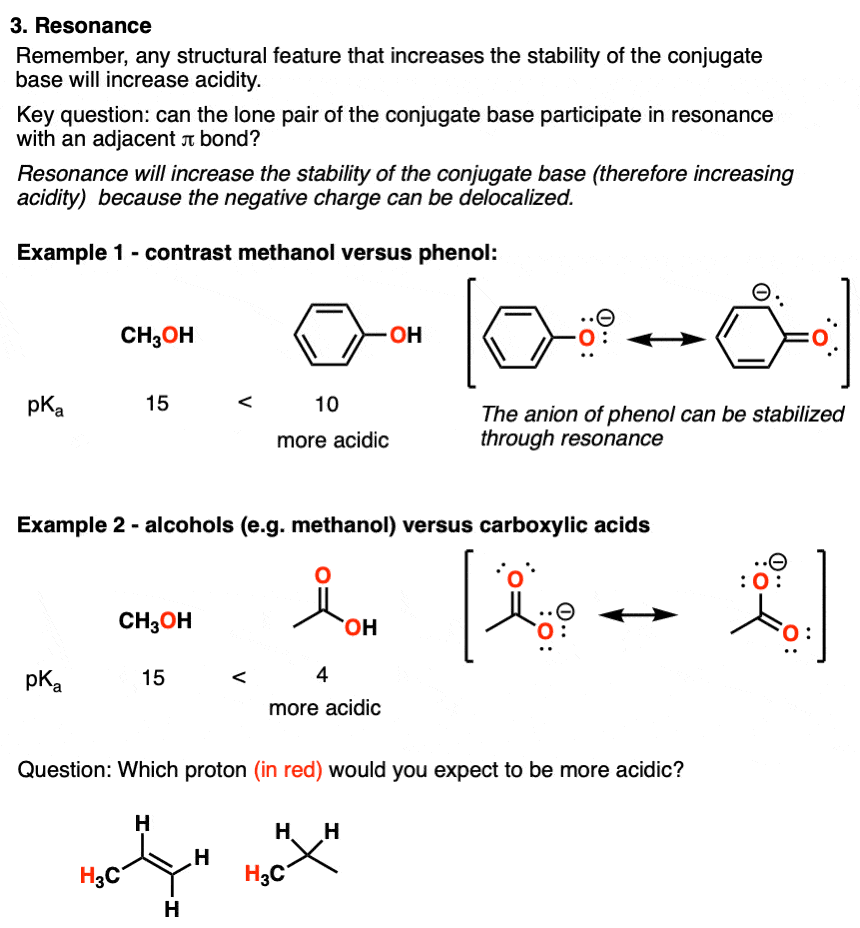
5 Key Factors That Influence Acidity In Organic Chemistry