
Microbiological Media Management - SOP & Guideline - Pharma Beginners
4.7 (737) In stock

4.7 (737) In stock
Standard Operating Procedure (SOP) and Guideline for the Receipt, Storage, Preparation, Growth Promotion Test, use, and Disposal of microbiological media.

Microbiological Media Management - SOP & Guideline - Pharma Beginners

🏆 Mastering SOP Content: Elevating Pharma Excellence 📊💡

Quality control in microbiology

PDF) Preparing SOP for Microbiology Laboratory: A Short Guideline

SOP for Microbial Limit Test- Detailed SOP
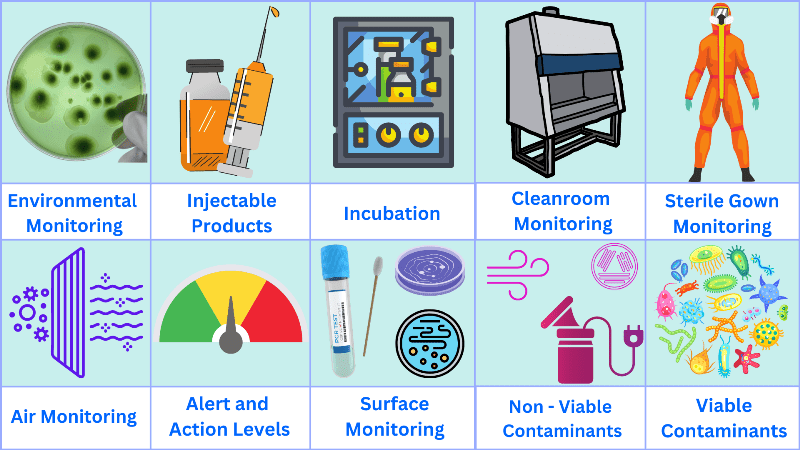
What is environmental monitoring in pharmaceutical industry

Update to EU GMP Chapter 6 - Quality Control - Inspired Pharma Training

SOP For Microbial Culture Management - Pharmaceutical Guidelines
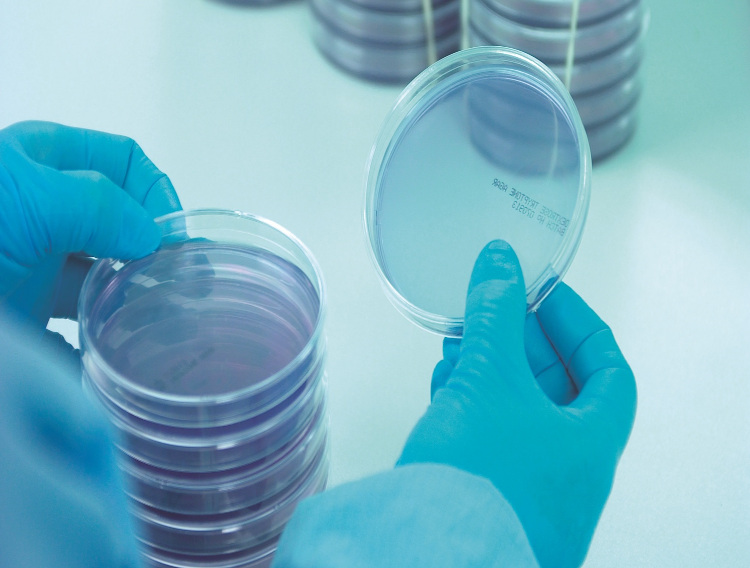
The importance of growth promotion testing
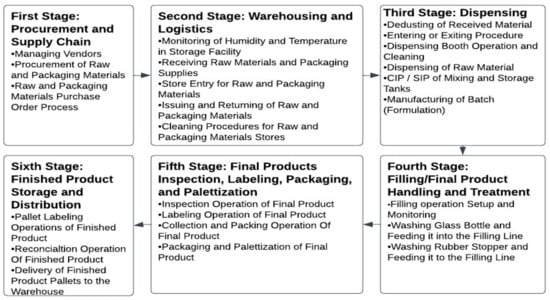
Sustainability, Free Full-Text

Culture techniques, procedures and regulations that should be included

PDF) Microbiological Culture Media: A Complete Guide for
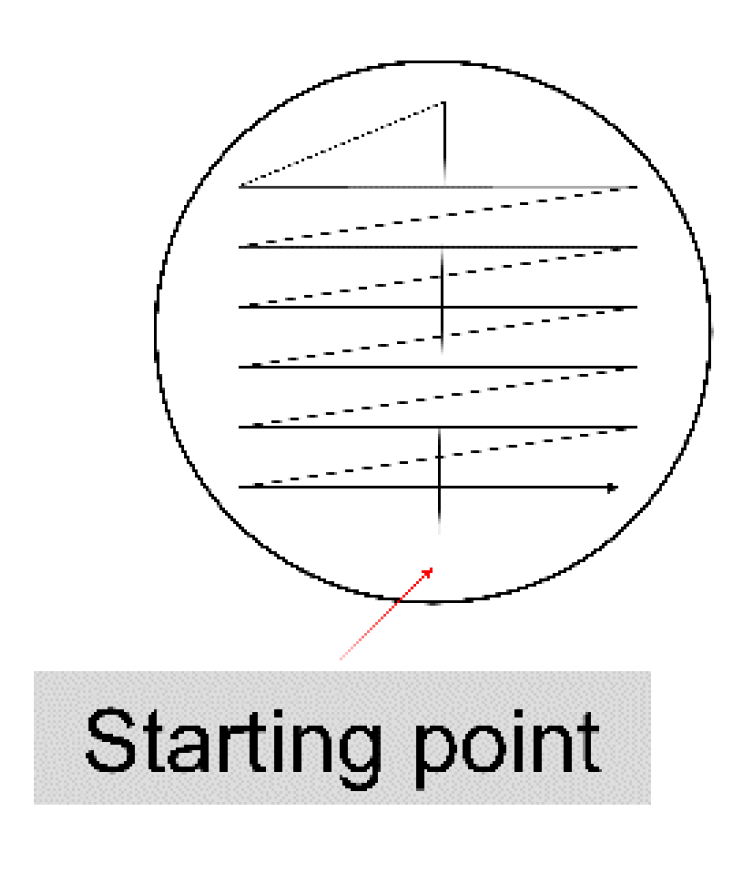
Diagnostics, Free Full-Text

Standard Operating Procedure Template - SOP Template