
How Do Instant Ice Packs Work?
4.9 (761) In stock

4.9 (761) In stock
Instant ice packs cool rapidly and provide soothing relief to injured areas, helping to reduce swelling and speed up recovery rates. But how do they work? We explain why they get cold, and the science behind the chemical reaction that makes them such a useful part of any first aid kit.

How to use an instant ice pack – Brenniston

Instant Ice Packs - How do they work? - Cleaning Supplies 2U
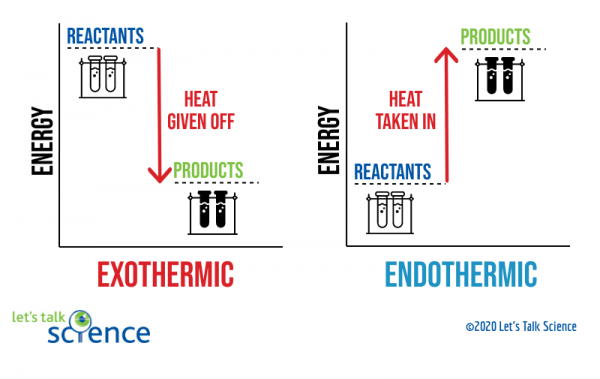
The Cold Pack: A Chilly Example of an Endothermic Reaction

What's The Difference Between Instant Cold Packs, Gel Packs, and Clay - IceWraps

Steroplast Reusable Clear Hot and Cold Pack, Instant Pain Relief

Instant Cold Pack - The Chemicals Inside - Product Breakdown

Instant Ice Packs(Temperature): Inside an instant ice pack, there

Instant Ice Packs: FAQs Answered

Koolpak Instant Ice Packs

Instant Ice Packs - How do they work? - Cleaning Supplies 2U
How to use an instant ice pack – Brenniston
Reactions and temperature changes - Exothermic and endothermic reactions - AQA - GCSE Combined Science Revision - AQA Trilogy - BBC Bitesize

Instant Cold Pack, XL Ice Packs (9 x 6) - Ready First Aid

How Do Instant Ice Packs Work?