Exploring Anodic & Cathodic Tafel Curves: Uneven Electrodeposition?
4.8 (649) In stock
4.8 (649) In stock
The shapes of the anodic and cathodic Tafel curves are different. What does it mean? Does it mean that the electrodeposition of the copper onto a surface of an electrode is uneven? If yes, I am also thinking that this has something to do with the macrothrowing power? Since it was done in an

Tafel plot for anodic and cathodic portions of the j versus η

High Speed Selective Jet Electrodeposition of Gold and Gold Alloys
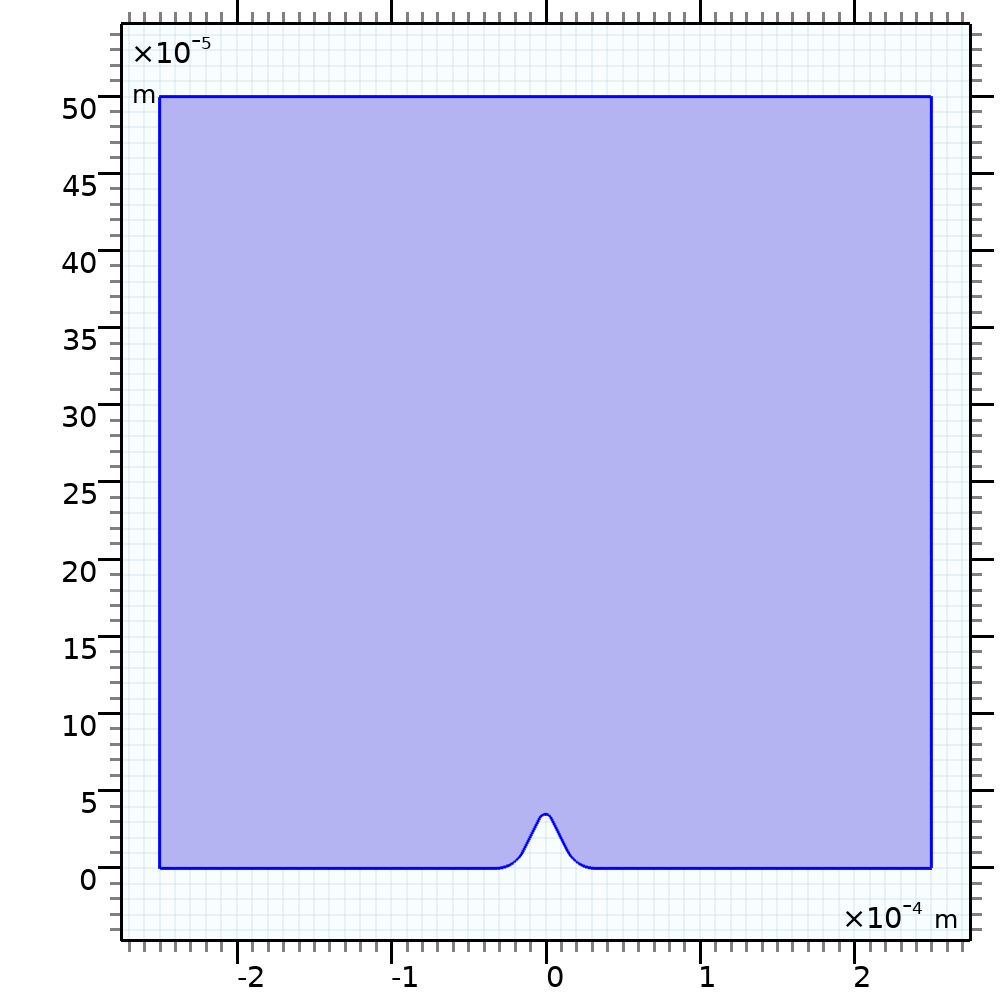
The Art and Science of Electrochemical Plating

Simulated Microgravity Accelerates Alloy Corrosion by Aspergillus

Superhydrophobic Surface and Lubricant‐Infused Surface
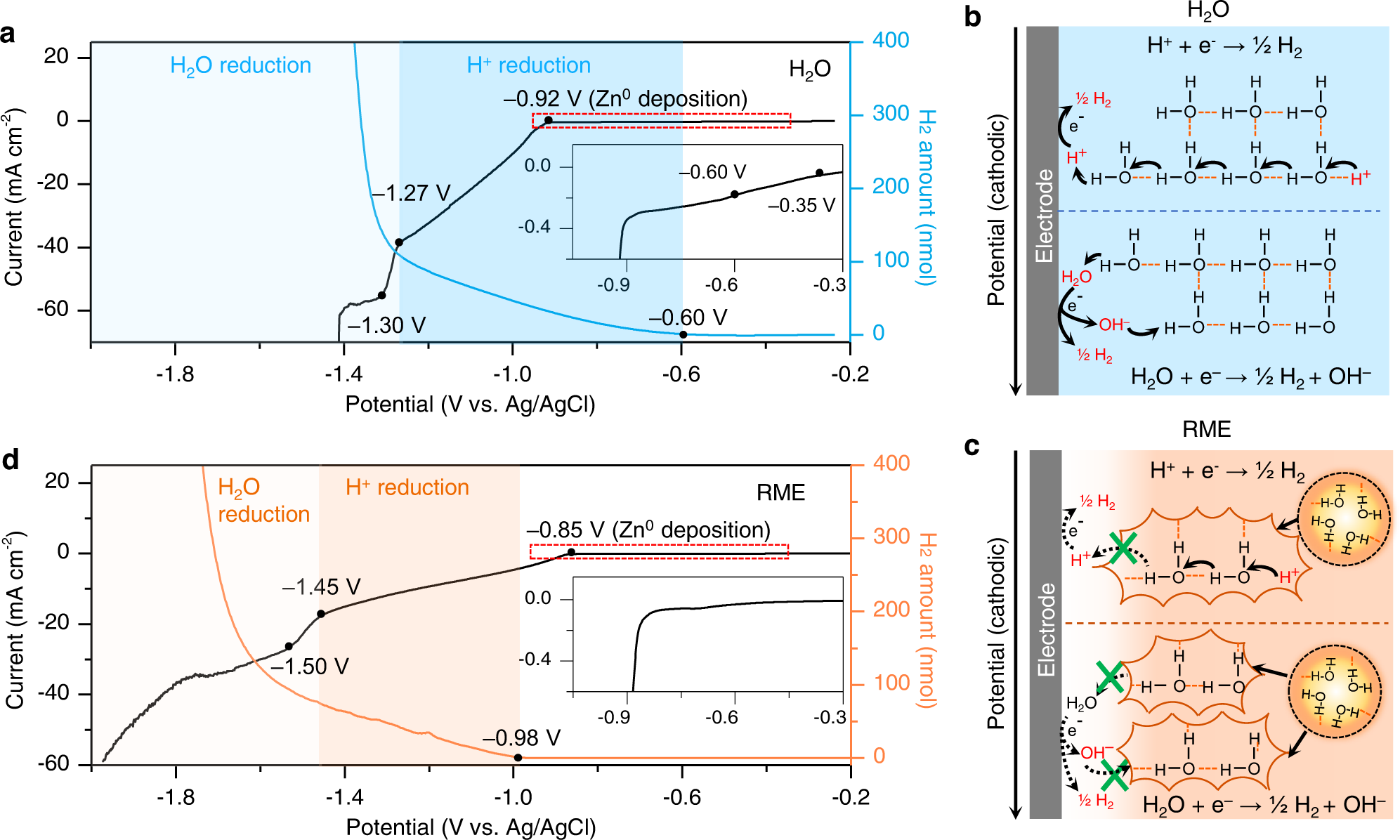
Sulfolane-containing aqueous electrolyte solutions for producing

Modification of surface hardness, wear resistance and corrosion

Strained carbon steel as a highly efficient catalyst for seawater

Ultrahigh‐Rate Zn Stripping and Plating by Capacitive Charge

Electrodeposition of Ni–Mo alloy coatings from choline chloride
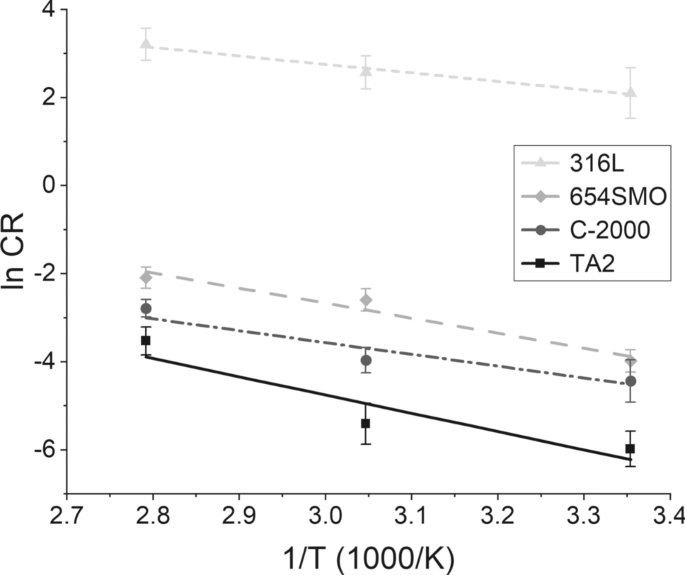
Performance-Based Selection of the Cathode Material for the
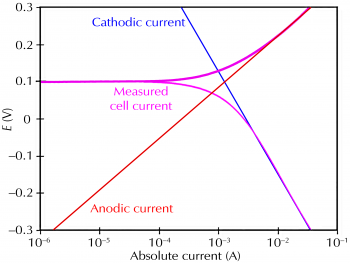
Electrochemical Corrosion Measurements-Galvanic Corrosion Gamry

PPT - Electrodeposition PowerPoint Presentation, free download

Electrochemical Deposition Toward Thin Films